1 approved prescription drug products with therapeutic equivalence evaluations. It is widely accepted as the authoritative source for determining therapeutic equivalence among multisource drug products.
The Orange Book Introduction.

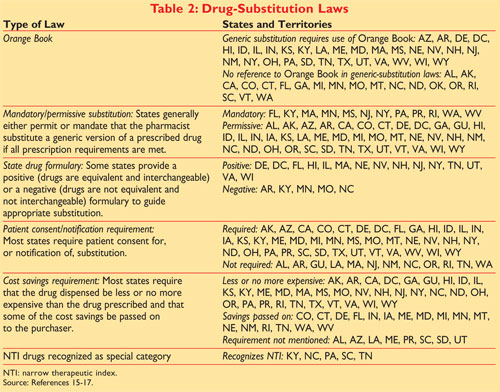
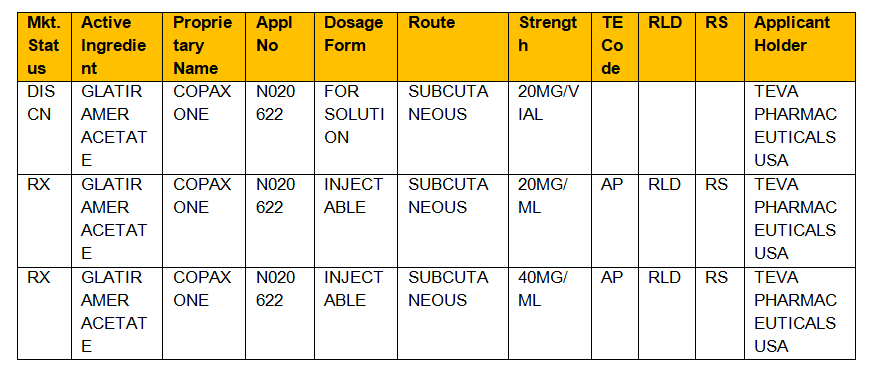
. CDR Kendra Stewart RPh PharmD. Then use the Ingredient Search for. In the electronic Orange Book a reference standard is identified by RS in the RS column.
R i s k r e. The orange book is published annually and the 2015 edition is 35th edition of orange book1 It is freely available for. Objectives What is a Generic Drug.
Type The group or category of approved drugs. Food and Drug Administration in both bound and electronic formats. First published in 1971 the original Orange Guide contained British Good Manufacturing Practice and was entitled Guide to Good Pharmaceutical Manufacturing Practice.
Use of unapproved drugs require complete CMC information depending on the nature of the study. FDAs Approved Drug Products with Therapeutic Equivalence Evaluations Orange Book identifies drug products approved on the basis of safety and effectiveness. The Orange book has been revised.
FDA orange book The official name of FDAs orange book is Approved Drug Products with Therapeutic Equivalence Evaluations. The FDA keeps a list known as the Orange Book of every approved therapeutic equivalent. Rules and Guidance for Pharmaceutical Manufacturers and Distributors commonly known as the Orange Guide brings together all the main European and UK directives regulations and legislation relating to the manufacture and distribution of medicines.
The FDAs list of Approved Drug Products with Therapeutic Equivalence Evaluations. Get emails about this page. The orange book is a list of generic drugs approved by FDA.
The Orange Book is an important publication published by the FDA that serves as the gold standard reference for generic drug substitution. Orange Book Approved Drug Products with Therapeutic Equivalence Evaluations Brand-generic therapeutic equivalencies Found in accessdatafdagov. The Orange Book Risk Management Principles.
This determines the ingredient s. The full publication title is Approved Drug Products with Therapeutic Equivalence Evaluations but it is commonly known as the Orange Book. The Orange Book is a compendium of significant unimplemented nonmonetary recommendations for improving departmental operations.
Not much more than 30 pages in length this voluntary guide was an aid to manufacturers to understand the needs of the regulatory authoritys requirements for the manufacture of. Medical Dictionary 2009 Farlex and Partners. The Orange Book is composed of four parts.
1 Generic substitution laws are state specific and many require use of the. Updated with Orange Book. First if you have the trade name search the Electronic Orange Books Rx or OTC section using the Proprietary Name search.
Basics in drug approval process with reference to the Orange Book Presented by. Rucha Pathak Roll No. Format is RX OTC DISCN.
The FDAs Orange Book identifies approved drug products. Office of Generic Drugs Policy Center for Drug Evaluation Research US. CSA 7395 7245 7498 1590 7025 7034 7011 7431 7031 7012 DEA SUBSTANCE NUMBER SCH NARC OTHER NAMES 4-Methyl-25-dimethoxyamphetamine I N DOM STP.
2 approved over-the-counter OTC drug products for those drugs that. Formally known as Approved Drug Products with Therapeutic Equivalence Evaluations the orange book lists drugs which are not only safe but also effective for human use. Approved Drug Products with Therapeutic Equivalence Evaluations.
The Orange Book formally titled Approved Drug Products With Therapeutic Equivalence Evaluations is a comprehensive list of approved drug products published by the FDA. The 2022 edition of Rules and Guidance for Pharmaceutical Manufacturers and Distributors the Orange Guide is now available through. Food and Drug Administration.
The Office of Inspector General. One prescription example would be combined oral contraception also. PCS - Dr Z.
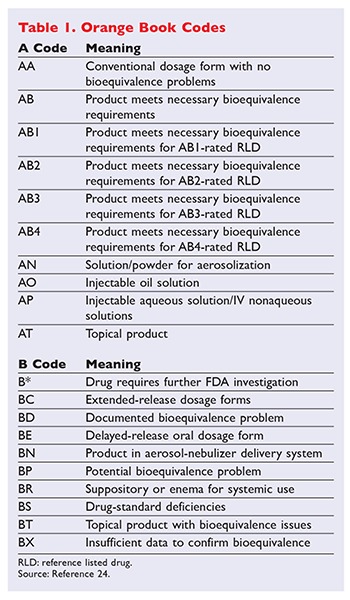
The Orange Book is a reference source that gives insight on whether or not two drugs have Therapeutic Equivalences. The Approved Drug Products with Therapeutic Equivalence Evaluations published by the US. FDA has draft guidance explaining that certain currently marketed drug ingredients were marketed before current FDA legislation.
Sets with similar terms. The cost that the patient pays at the pharmacy point of sale. Originally this book was published in October 1980 with orange cover and thus the name orange book.
G o v e r n a n c e and L e a d e r s I n te g ra o n h i p C o l a b or ti o n Information Insight Insight Information Communication. Typically refers to a drug product that can be purchased without a prescription. Sponsors using these products should consult FDA about the need for an IND.

The Introduction Of An Orange Book

The Introduction Of An Orange Book

The Introduction Of An Orange Book

The Introduction Of An Orange Book

Therapeutic Equivalence Codes Effects Substitution Video Lesson Transcript Study Com

Investigational New Drug Orange Book Understanding On 505 B 2 A

Pharmaceutical Press Rules And Guidance For Pharmaceutical Manufacturers And Distributors 2022 The Mhra Orange Guide

The Introduction Of An Orange Book

The Introduction Of An Orange Book

List Of Branches Of Science And Definition Classification Of Science Branches Of Science Biology Facts General Knowledge Facts
Regulatory 101 Drug Name Modifiers Definition Categories Generics And Capa Ivt
Orange Book And Its Applications Legal Advantage

The Introduction Of An Orange Book
Appendix A Medical Terminology And Abbreviations In Manual For Pharmacy Technicians
/doctor-826e0c116cd549d98e327f1184c622d9.jpg)